https://doi.org/10.1140/epjs/s11734-026-02220-2
Regular Article
Synthesis, crystal structure elucidation, and molecular interaction studies of a guanidinium naphthoate adduct via Hirshfeld and docking analyses
1
PSG Institute of Medical Science and Research, Coimbatore, Tamil Nadu, India
2
Department of Chemistry, PSGR Krishnammal College for Women, 6410014, Coimbatore, Tamil Nadu, India
3
Glyco-Chemistry Laboratory, School of Physical Sciences, Jawaharlal Nehru University, 110067, New Delhi, India
4
Department of Biotechnology, Indira Gandhi National Tribal University, 484887, Amarkantak, Madhya Pradesh, India
5
Graduate School of Life Science and Systems Engineering, Kyushu Institute of Technology, 2-4 Hibikino, Wakamatsu, 808-0196, Kitakyushu, Japan
6
Department of Chemistry, Institute of Science, Banaras Hindu University, 221005, Varanasi, Uttar Pradesh, India
a
This email address is being protected from spambots. You need JavaScript enabled to view it.
b
This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
26
November
2025
Accepted:
17
February
2026
Published online:
9
March
2026
Abstract
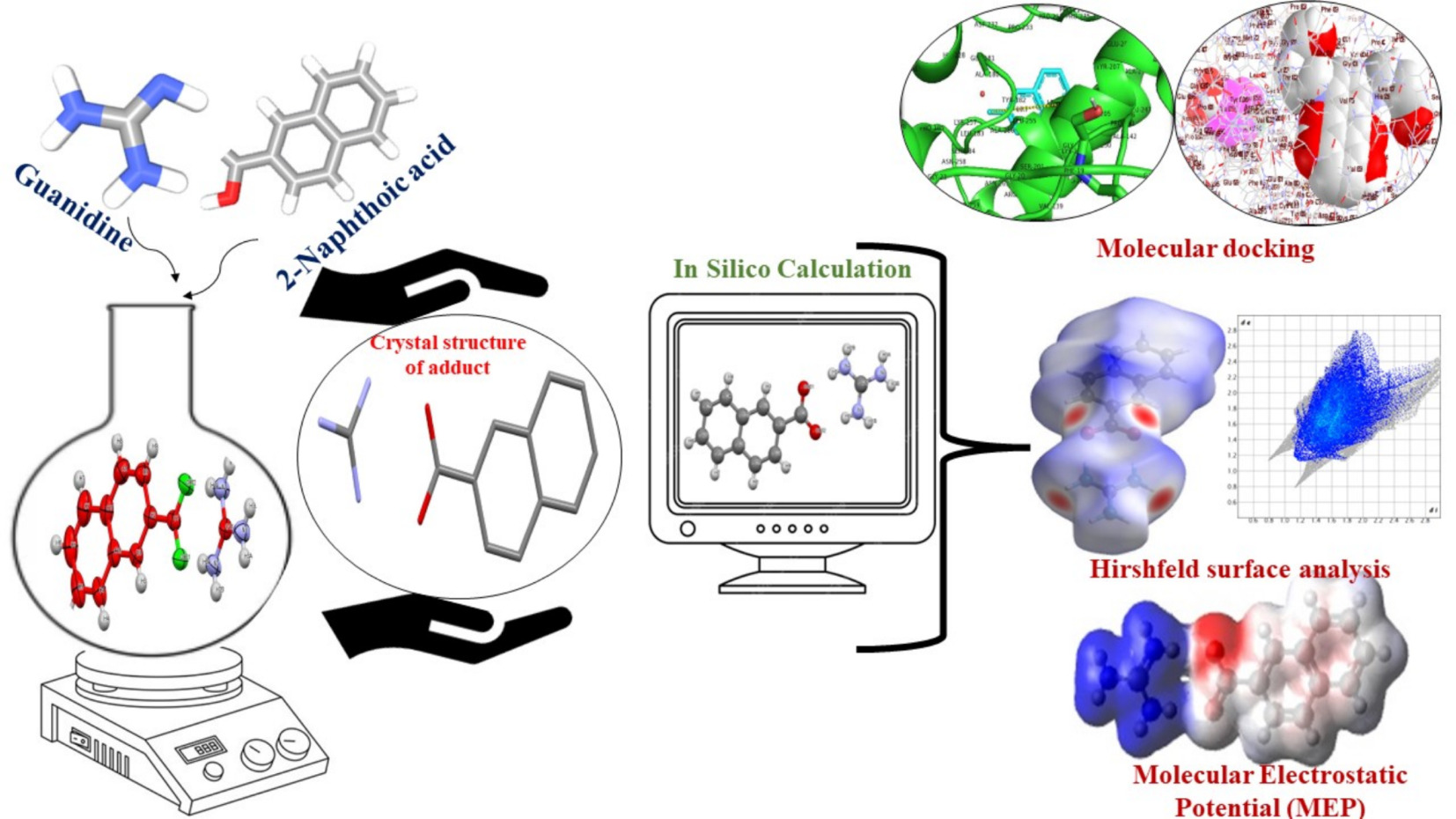
A new guanidinium naphthoate adduct in the form of a single crystal was established by adopting a slow evaporation growth technique with a suitable solvent. The bonding interactions, the presence of functional groups, and thermal stability were confirmed using various characterization techniques. The synthesized transparent compound crystallizes in a monoclinic lattice and exhibits centrosymmetric space-group symmetry P1 21/c1. Hirshfeld surface analysis and DFT calculation were performed to investigate the molecular interactions in the crystal. The dependencies of the extinction coefficient and skin depth on photon energy were illustrated. The slope for non-spontaneous, endothermic, and slow reactions was found to be 0.88–0.99 at different temperatures. The abundance of H–H and O–H/H–O interactions reveals that both van der Waals interactions and hydrogen bonding are the primary driving forces in the crystal packing. The blood–brain barrier (BBB) value is 0.46, indicating that the compound can cross the BBB and exhibit biological activity within the central nervous system. The compound demonstrates 95.53% absorption in the human gastrointestinal tract, suggesting good oral bioavailability.
Copyright comment Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
© The Author(s), under exclusive licence to EDP Sciences, Springer-Verlag GmbH Germany, part of Springer Nature 2026
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.